Clay Classification: Navigating The Spectrum Of Options
Clay minerals are an important component of soils and have significant effects on soil properties and behaviors. Clay particles are classified into groups based on their crystalline structure, chemical composition, and other characteristics. Proper classification of clays is crucial for understanding soil mechanics and engineering properties, assessing soil fertility, and selecting appropriate construction materials. The main categories of clay minerals include kaolinite, smectite, illite, chlorite, vermiculite, and palygorskite. There are also mixed layer clays that contain interstratified combinations of these mineral types.
Classification allows us to correlate specific clay types with stability, shrink-swell potential, cation exchange capacity, and other attributes. For instance, smectite clays like montmorillonite have a higher shrink-swell potential than kaolinite clays. Accurate identification informs agricultural management, hazard mitigation, and geotechnical design. As we will explore in this article, the spectrum of clay mineral groups ranges from relatively inactive kaolinites to highly reactive and expansive smectites. Understanding where a given clay falls on this spectrum is essential.
Kaolinite Clays
Kaolinite is the most common clay mineral belonging to the kaolin group ([https://geologyscience.com/minerals/kaolinite/?amp]). It is a 1:1 layered silicate composed of one tetrahedral silica sheet and one octahedral alumina sheet. The chemical structure of kaolinite is Al2Si2O5(OH)4. It is a soft, earthy, usually white mineral produced by the chemical weathering of aluminum silicate minerals such as feldspar ([https://activeminerals.com/blog/kaolin-guide/]).
Some key properties of kaolinite clays include:
- Low plasticity and sticky when wet
- Poor dry strength with low cohesion
- Smooth and silk feel
- Low cation exchange capacity (CEC)
Kaolinite clays are commonly found in kaolin and ball clays. They have various uses including in ceramics, paper coating and filling, refractories, fiberglass, paint, rubber, and catalysis due to their high fusion temperature, chemical inertness, whiteness, and fine particle size ([https://activeminerals.com/blog/kaolin-guide/]). Major sources of kaolinite clays are the kaolin pits in Georgia, USA and the china clay quarries in Cornwall, England.
Smectite Clays
Smectite clays, like montmorillonite, are characterized by their expandability and ability to swell with the addition of water. They have a net negative charge that is balanced by cations like Na+, Ca2+, Mg2+, and H+ adsorbed on interlayer surfaces (Odom, 1984). The weak Van der Waals forces between layers allow water molecules and cations to enter between the layers, forcing the layers apart and allowing smectite clays to expand to up to 20 times their original volume.
The most common smectite clay is montmorillonite, which is the main constituent of bentonite. Montmorillonite forms from the alteration of volcanic ash and tuff in marine environments and has a high cation exchange capacity due to isomorphous substitution. It is used commercially for its swelling, adsorption, and cation exchange properties in products like cat litter, cosmetics, pharmaceuticals, and drilling fluids (Wikipedia). Other smectite group minerals include beidellite, nontronite, saponite, and hectorite.
The properties of smectite clays make them useful as adsorbents, binders, thickeners, and fillers across many industries. Their ability to expand allows them to absorb large quantities of water and other molecules between layers. The charge deficiency from isomorphous substitution gives smectites a high cation exchange capacity, allowing them to adsorb and exchange cations. When hydrated, the increased viscosity and plasticity allow smectites to act as effective thickeners, suspending agents, and binders.
Illite Clays
Illite clays are micaceous minerals comprising layered alumino-silicates. They contain more water and less potassium than true micas, but have a mica-like sheet structure. Illite is poorly crystallized compared to other clay minerals like kaolinite. The properties of illite arise from its platy structure consisting of silica tetrahedral sheets layered between alumina octahedral sheets (Britannica, 2022).
Some key properties and uses of illite clays include:
- Moderate cation exchange capacity of 10-40 meq/100g due to some expandability between layers (Wikipedia, 2022). This allows illite to absorb certain cations like potassium and ammonium.
- Relatively low plasticity compared to smectite clays.
- Illite rich clays are often used for manufacturing bricks, ceramics, and cements.
- The fine particle size of illite makes it useful as a filler material in papers, paints, rubbers and plastics.
Illite clays have some similarities with smectite clays in their sheet silicate structure. However, illite has much less swelling and lower cation exchange capacity than smectites. The key difference is that illite layers are held together too rigidly to allow for much interlayer expansion with water. In contrast, smectites have expandable interlayers that take up water and cations (Wikipedia, 2022).
Chlorite Clays
Chlorite clays are a group of phyllosilicate minerals characterized by their green color and perfect cleavage. Some key properties of chlorite clays include:
- Platy, micaceous texture – chlorite minerals tend to occur as thin, flexible flakes or scales.
- Green, blue-green, or gray-green color – caused by the presence of iron and magnesium.
- Perfect cleavage – chlorite minerals break evenly along one direction, like mica.
- Low hardness – typically 2-3 on the Mohs hardness scale.
- Greasy or pearly luster.
The most common chlorite mineral is clinochlore. Other chlorite group minerals include chamosite, nimite, and pennantite. Chlorite clays are used in a variety of applications including as a source of magnesium and iron, as a cosmetic ingredient, and as an industrial absorbent (source). Major sources of chlorite clays include Brazil, Finland, India, and the United States.
Vermiculite and Palygorskite
Vermiculite is a 2:1 clay mineral similar to micas that exhibits the unique property of expanding greatly when heated. The heating process causes water molecules lodged between the vermiculite’s platelets to convert to steam and push the plates apart. This expansion creates chambers that give vermiculite its extremely lightweight and absorbent nature (Encyclopedia Britannica).
The expanded version of vermiculite has a variety of specialized uses, including as a soil additive, packaging material, insulation, and absorbent for hazardous spills. Raw vermiculite is mined and then processed via heating to obtain the expanded version. Major sources of vermiculite are found in South Africa, China, Brazil, and the United States (Kumari, 2021).
Palygorskite, also known as attapulgite, is a clay mineral with a fibrous or chain-like morphology. Its unique structure gives palygorskite useful adsorbent and decolorizing properties. Palygorskite is primarily used as an additive in a variety of industrial and consumer products where it can thicken, suspend particles, increase viscosity, and adsorb impurities (ScienceDirect).
Palygorskite deposits are found in regions such as Georgia, Florida, and Africa. Extensive processing is required to prepare the raw palygorskite into purified grades for commercial use.
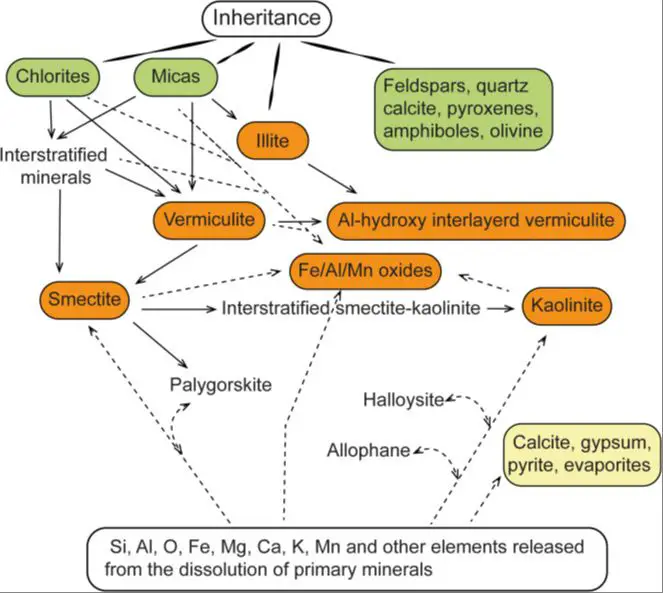
Mixed Layer Clays
Mixed layer clays are composed of two or more different types of clay minerals stacked together in an alternating fashion. They form where one type of clay mineral transforms into another type due to changes in environmental conditions such as temperature, pressure, and chemical composition of waters interacting with the minerals.
The most common types of mixed layer clays include:
- Illite-smectite
- Chlorite-smectite
- Chlorite-vermiculite (corrensite)
- Kaolinite-smectite
- Illite-vermiculite
As one clay mineral transforms into another, their structures are interlayered resulting in a mixed layer clay. The specific crystal structure and properties of the mixed layer clay depends on the relative proportions of the constituent minerals. X-ray diffraction analysis can identify and quantify the different mineral layers present.
Factors like changes in climate, burial depth, and exposure to hydrothermal fluids drive the transformation process from one clay mineral to another over geologic timescales. Mixed layer clays illustrate the dynamic nature of clay mineral formations.
Factors Affecting Properties
The properties of clay are influenced by several key factors including mineralogy, particle size, and exchangeable ions. According to an article on Springer Link, “Factors Affecting Clay Formation,” the type of minerals present has a significant effect on clay properties (https://link.springer.com/article/10.1346/CCMN.1957.0060110). The primary clay minerals that impact properties are kaolinite, smectite, illite, and chlorite. Kaolinite clays tend to have low plasticity and shrink-swell capacity. Smectite clays exhibit high cation exchange capacity and expansion. Illite and chlorite have intermediate properties.
Particle size also affects clay behavior as described in the IntechOpen chapter, “Basics of Clay Minerals and Their Characteristic Properties” (https://www.intechopen.com/chapters/76780). Finer clay particles have higher surface area and reactivity. As particle size decreases, plasticity and cohesion increase. The type and amount of exchangeable cations further modify clay properties by influencing flocculation, permeability, and dispersion.
Testing and Analysis
There are various methods for testing and analyzing clays to determine their properties and suitability for different applications. Some common testing methods include:
Particle Size Analysis – This involves passing the clay sample through a series of sieves with progressively smaller mesh sizes to determine the size distribution of particles. This helps classify clays into coarse, medium and fine fractions.
Atterberg Limits – The liquid limit, plastic limit, and plasticity index are determined. This helps assess the plasticity and workability of the clay. Standard test procedures are defined by ASTM D4318.
X-Ray Diffraction – XRD analysis identifies the clay mineral types present by measuring diffraction patterns when x-rays interact with the crystalline structure of clays. This helps identify the specific clay species.
Thermal Analysis – Techniques like thermogravimetric analysis (TGA) and differential scanning calorimetry (DSC) characterize clays by studying their behavior on heating. This provides data on properties like loss on ignition, transformations, and reaction temperatures.
Chemical Analysis – Elemental analysis determines the chemical composition in terms of oxides. This quantifies the levels of silica, alumina, iron, alkalis and other oxides present.
Rheological Testing – Viscometers and rheometers measure viscosity, yield stress and other rheological parameters important for processing and applications.
Proper testing and analysis is key to understanding the properties and behavior of different clays. The data obtained guides effective selection and use of clays.
Conclusion
In summary, understanding the classification and properties of different clay types is crucial for many applications and industries. The key learnings are that clays can be categorized into major groups based on their mineralogy and plasticity, with kaolinite, smectite, illite and chlorite being the main types. Classification testing analyzes a clay’s Atterberg Limits, cation exchange capacity, thermal analyses and X-ray diffraction patterns. Each clay type has unique characteristics – kaolinites have low plasticity and shrinkage, smectites swell and shrink considerably, illites have moderate properties, while chlorites and vermiculites have higher charge densities. Knowledge of a clay’s specific properties allows civil engineers to assess soil stability for construction projects, helps determine agricultural uses for different soils, enables manufacturers to select the right clay for products, and helps geologists interpret sites based on the clay minerals present. Proper clay identification provides scientists, engineers and builders with critical data to successfully apply these very important minerals in a wide range of industries and products.